-
Previous paper
Wetland and benthic cover changes in Moreton Bay
Eva M. Kovacs1 Hannah L. Tibbetts2, Simon Baltais3, Mitch Lyons1, Jennifer Loder4, 5 and Chris Roelfsema1. -
This paper
The impact of marine pollutants and marine debris in Moreton Bay
Kathy A. Townsend1,2, Christine Baduel3, Vicki Hall4, Jennifer Loder5, Veronica Matthews6, Jochen Mueller3, Rachael Nasplezes7, Qamar Schuyler8, Heidi Taylor9, Jason van de Merwe10, C. Aleander Villa3 and Liesbeth Weijs3, 10 -
Next paper
Projected changes to population, climate, sea-level and ecosystems
Megan I. Saunders1,2, Rebecca Runting3, Elin Charles-Edwards2, Jozef Syktus4 and Javier Leon5
The impact of marine pollutants and marine debris in Moreton Bay
Authors
Kathy A. Townsend1,2, Christine Baduel3, Vicki Hall4, Jennifer Loder5, Veronica Matthews6, Jochen Mueller3, Rachael Nasplezes7, Qamar Schuyler8, Heidi Taylor9, Jason van de Merwe10, C. Aleander Villa3 and Liesbeth Weijs3, 10Author affiliations
- School of Science and Engineering, University of the Sunshine Coast, Fraser Coast Campus, 4655, Australia;
- School of Biomedical Sciences, The University of Queensland, St Lucia Qld, 4072, Australia;
- Queensland Alliance for Environmental Health Science, The University of Queensland, St Lucia Qld, 4072, Australia;
- Conservation and Sustainability Services, Department of Environment and Science, Queensland Government, Brisbane Qld, 4000, Australia;
- Reef Check Australia, Brisbane Qld, 4000;
- Menzies School of Health Research, Casuarina NT, 0811, Australia;
- Healthy Waterways and Catchments, Brisbane Qld, 4000, Australia
- CSIRO Marine and Atmospheric Research, Tas., 7000, Australia;
- Tangaroa Blue, Qld, 4877, Australia
- Australian Rivers Institute, Griffith University, Nathan, Qld, 4111
Corresponding author
ktownse1@usc.edu.auORCID
Kathy Townsend: https://orcid.org/0000-0002-2581-2158
Jennifer Loder: https://orcid.org/0000-0002-8460-7162
Veronica Matthews: https://orcid.org/0000-0002-1319-257X
Jochen Mueller: https://orcid.org/0000-0002-0000-1973
Rachael Nasplezes: https://orcid.org/0000-0002-5600-6036
Qamar Schuyler: https://orcid.org/0000-0002-2473-4311
Jason van de Merwe: https://orcid.org/0000-0002-0838-5780
Alexander Villa: https://orcid.org/0000-0001-7043-9746
Liesbeth Weijs: https://orcid.org/0000-0002-2399-1711
Book
The impact of marine pollutants and marine debris in Moreton Bay
Chapter
Research Paper Title
The impact of marine pollutants and marine debris in Moreton Bay
Cite this paper as:
Townsend K, Baduel C, Hall V, Loder L, Matthews V, Mueller J, Nasplezes R, Schuyler Q, Taylor H, van de Merwe J, Villa CA, Weijs L . 2019. The impact of marine pollutants and marine debris in Moreton Bay. In Tibbetts, I.R., Rothlisberg, P.C., Neil, D.T., Homburg, T.A., Brewer, D.T., & Arthington, A.H. (Editors). Moreton Bay Quandamooka & Catchment: Past, present, and future. The Moreton Bay Foundation. Brisbane, Australia. Available from: https://moretonbayfoundation.org/
DOI
10.6084/m9.figshare.8073851
ISBN
978-0-6486690-0-5
Abstract
Moreton Bay is affected by a wide range of persistent pollutants, including the following four broad groups: (i) dioxins and persistent organic pollutants (POPs), (ii) trace elements (e.g. metals, metalloids and non-metals), (iii) perfluoroalkyl substances (PFAAs) and (iv) plastic-based marine debris. The pollutants discussed in this paper come from diverse chemical groups, and are similar in that they are persistent in the environment and impact the health of animals and/or humans. While most of these pollutants are banned and/or actively monitored by health organisations on a global scale, (e.g. PCBs and DDT) many are still entering our waterways daily (e.g. marine debris). We discuss how dioxins have impacted on the health, lifestyle, and culture of the region’s Traditional Custodians. Case studies are presented, highlighting the impacts of the four pollutant groups on marine megafauna found in Moreton Bay including sea turtles, seabirds, and stingrays. In all cases, the authors recommend increased monitoring and the development of new strategies to reduce the four pollutant groups entering Moreton Bay.
Keywords: marine rubbish, polymer, PCB, DDT, PFAA, heavy metals, human impact
Introduction
The term marine pollution brings to mind many different images ranging from beaches covered in oil through to waterways filled with plastic bags. While the authors acknowledge that there are many types of pollution, including pharmaceuticals, petrochemicals, nutrients and sedimentation, this paper focuses on four persistent pollutant groups currently impacting Moreton Bay (Quandamooka): persistent organic pollutants (POPs), dioxins, trace elements, perfluoroalkyl substances (PFAAs) and marine debris.
Moreton Bay (Quandamooka) is situated off the coast of one of the largest cities in Australia, Brisbane. Colonised in 1824 as a penal colony (1) and with a current population of over one million (2), managing man-made waste has long been a challenge. Prior to European colonisation, pollution was primarily organic-based, including items such as food scraps, human waste, and stormwater. However, the pollutants being introduced to Moreton Bay changed as Brisbane grew as a city. For example, pharmaceuticals, sediment and nutrient inputs have all increased as the population has grown. The four persistent pollutant groups reviewed in this paper have one thing in common—they are all highly resistant to degradation/decay processes. As such, all of these compounds bioaccumulate in aquatic organisms and many can biomagnify up the food chain, further exacerbating their long-term environmental impact. We review how they have changed over time and discuss how POPs have impacted on the health, lifestyle, and culture of the region’s Traditional Custodians. We present case studies highlighting the impacts of these key pollutants on marine megafauna including sea turtles, seabirds and stingrays found in Moreton Bay. We discuss the role that government agencies, non-government organisations and volunteer organisations have in monitoring and cataloguing marine pollution within the region. Although progress has been made in reducing marine pollution there are still many challenges to address. The paper concludes by suggesting strategies for improving pollutant management in Moreton Bay.
Persistent organic pollutants
Persistent organic pollutants (POPs) are hazardous to humans and wildlife and encompass a wide variety of chemicals with various structures and applications (3). The most toxic POPs are subject to the Stockholm Convention, which aims to reduce or eliminate the release of these pollutants on an international scale (4). They include chemicals that are now banned or restricted (e.g. PCBs, DDT), some that are released unintentionally (e.g. dioxins), and emerging new compounds that are still being used (e.g. chlorinated paraffins). These chemicals are highly lipophilic and persistent, so even banned POPs are still present in the environment and can be stored in soil, sediments and biota for decades to centuries (5). From these environmental sources, the chemicals can bioaccumulate to elevated levels, particularly in fatty tissues, and can biomagnify through the food chain, which explains why higher trophic organisms typically contain the highest levels of POPs (3, 6). Studies have shown that even low-level chronic exposure can lead to a wide range of adverse effects including cancer, reproductive and developmental effects, disruption to the immune system and damage to the nervous system for both humans (7) and marine species (6, 8, 9).
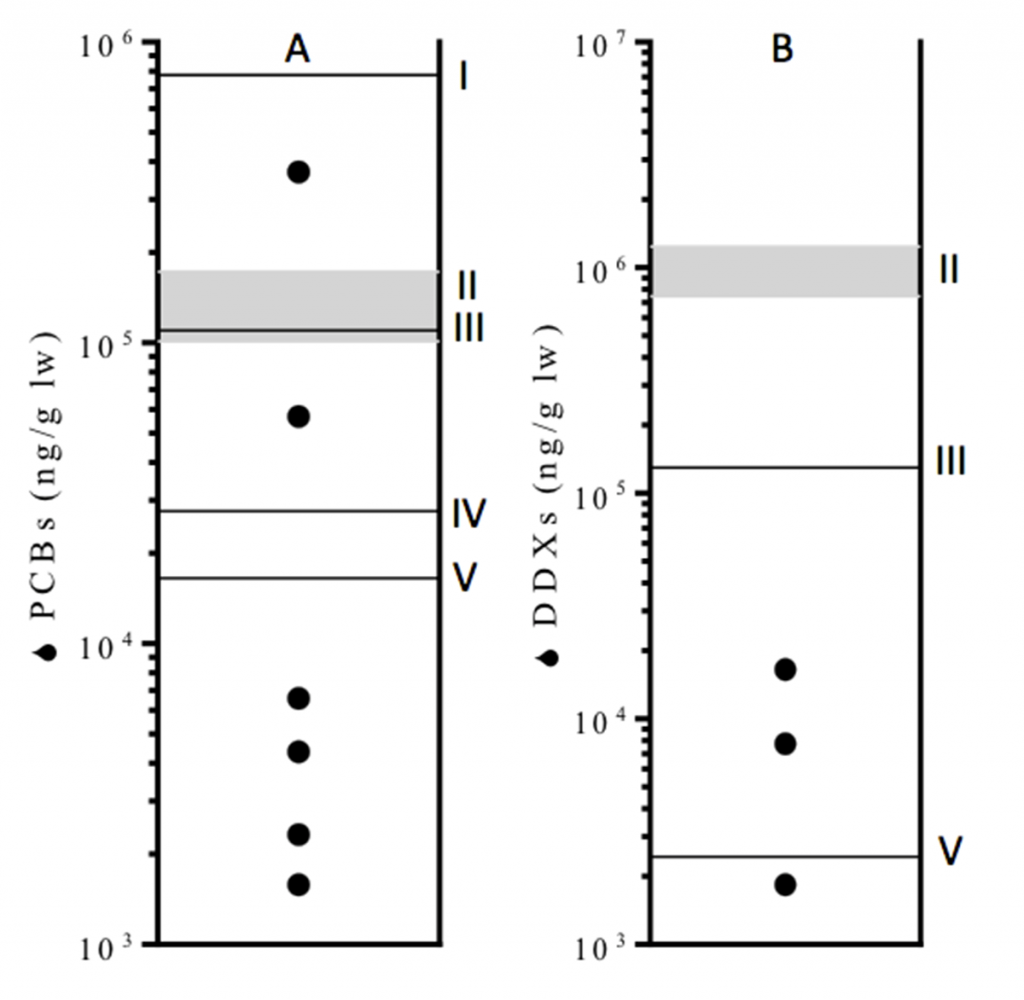
Several studies have investigated the sources, transport and levels of POPs in the Moreton Bay region (e.g. 10-13). Based on initial findings, dioxins (a chemical subset of POPs) were the focus of further investigations due to their ubiquitous and elevated levels in soils, sediments, seafood and other marine wildlife. In addition, PCBs and DDTs were found to be elevated in localised hotspots, especially in western Bay areas and in species frequenting these waters such as the humpback dolphin, Sousa sahulensis (Fig. 1) (14).
Case study 1: POPs and the Quandamooka people
Approximately 90% of the total exposure of humans to POPs occurs through intake of contaminated food, particularly lipid-rich products, including seafood. As a signatory to the Stockholm Convention, Australia funded a four-year National Dioxins Program in 2001 to increase knowledge of levels in the environment and determine the risk posed to the health of the population. As part of this assessment, toxic equivalent (or TEQ) levels in a number of retail products (including seafood) were analysed and human consumption levels were estimated from national surveys. The study reported that the TEQ levels of POPs in Moreton Bay seafood were 9 times higher in wild caught Australian marine/estuarine fish and 25 fold higher in retail fish compared to background levels measured for the National Dioxins Program (12). However, the TEQ levels of most seafood analysed were below the European Union (EU) guideline limits (11). The study concluded that the levels were found to be comparable to those measured in other urban areas, which similarly received historical discharges of agricultural, municipal and industrial wastewater. The conclusion from the program was that the risk to the general Australian population from dioxins was low (15).
However, the National Dioxins Program did not take into account population groups who may consume more self-caught seafood compared to the general population. Unique subsistence patterns of coastal and Indigenous populations may place them at greater risk of exposure to dioxins due to reliance on contaminated food sources (16, 17). The Quandamooka people of Moreton Bay have cared for the local environment for thousands of years (18). Their cultural practices ensured a sustainable abundant supply of seafood and other resources. Like other Indigenous communities, Quandamooka people would utilise most aspects of the catch, including lipid-rich organs such as roe and testes (melts). Dugong (Dugong dugon) was hunted for both its flesh and oil. Hunting practices played an important role in social cohesion as every part would be shared amongst families (18). Fishing and traditional hunting are an important affirmation of cultural identity and continue to be an essential aspect of Quandamooka life. These food sources have also been assumed to be a healthy and affordable dietary source.
Given the higher contamination levels in Moreton Bay seafood compared to retail food and the subsistence patterns of local communities, a health risk assessment was conducted for the Quandamooka and broader North Stradbroke Island community (19). Surveys showed this community consumed on average two to six times more seafood than the general Australian population. Depending on the level of seafood consumption, the exposure assessment found that the average monthly dioxin intake for the community ranged between 34 to 107 picogram TEQ/kg body weight/month—an order of magnitude higher than that estimated for the general population. Between 11 and 44% of the community may be exceeding the WHO safe intake guideline (70 picogram TEQ/kg body weight/month) at chronic exposure levels.
A delicate balance is required between educating the Quandamooka community about risks from consuming seafood compared to the nutritional and economic benefits, as well as the cultural benefits of maintaining consumption levels. Risk management strategies have been suggested that can effectively reduce contaminant intake, while having a low impact on beneficial aspects of seafood consumption (19). Some strategies include removing skin (and associated fat layers) from fish before cooking and eating more of the leaner fish species, such as snapper, whiting and flathead. The current study showed that local seafood consumption in coastal communities can result in high exposure to dioxins. It highlights the importance of considering local economic, cultural, and environmental conditions and information on contaminant fate processes when conducting human exposure evaluations.
Trace elements
Trace elements enter the marine environment in several ways including, weathering and erosion of rocks, atmospheric deposition and through industrial and domestic activities in the water and adjacent catchments. Trace elements can bind to both mineral particles and organic matter in the water column and settle out to accumulate in the sediment (20). When disturbed, or if water quality is altered, sediment-bound trace elements can be liberated and become bioavailable (21). Rainfall and flooding events can result in short-term enrichment and bioavailability (22, 23). Excessive accumulation of trace elements, even those essential for life, can be toxic to marine flora and fauna (24, 25).
Trace elements have been well documented in the waters (22, 26), sediments (27, 28, 29), flora (30) and fauna (14, 31, 32) of Moreton Bay. They are generally highest in the western Bay (31, 32), and are closely linked to point sources such as shipping/boating (22, 28) and refuse tips (33), as well as more generally to catchment land use (26, 27, 29). Sediment coring within the Bay shows an increasing concentration trend since the 1920s, likely due to industrial, agricultural and urban development in the adjacent catchments (34). Recently reported concentrations of several trace elements in western Bay surface sediments exceeded background reference ranges with arsenic (As), cadmium (Cd), cobalt (Co), copper (Cu), nickel (Ni), lead (Pb), and zinc (Zn) posing a medium to high risk to benthic biota (27).
Water quality guidelines, values and objectives for managing trace elements in Moreton Bay are outlined by the Queensland Government (35). However, these guideline levels are not consistent across the Bay, with some regions considered to be of ‘high ecological value’ (HEV), receiving 99% protection values in the ANZECC, while others, such as the western part of the Bay, are considered to be so impacted that they are no longer considered to be of HEV, and will only then be protected under the 95% guideline value (35, 36). It is important that researchers communicate with managers to regularly review the levels of protection and their trigger values to determine if they are adequate for protecting the Bay and its marine inhabitants.
Case study 2: Trace elements and sea turtles
Sea turtles are exposed to trace elements when they consume contaminated food sources such as seagrass, macroalgae, benthic crustaceans, soft sponges/corals and jellyfish, as well as when they incidentally ingest contaminated sediments during benthic feeding. Various trace elements were identified within tissues of stranded Moreton Bay sea turtles (Chelonia mydas, Caretta caretta, Eretmochelys imbricata, and Lepidochelys olivacea from 1990–91) at lower concentrations relative to other reported populations outside of the Bay, with the exception of cadmium (37). A subsequent investigation of stranded Moreton Bay green sea turtles (C. mydas) from 2006–07 described much higher cadmium and arsenic tissue concentrations than previously reported (38).
Interpreting the consequences of elevated trace element concentrations in sea turtles is difficult in the absence of any baseline data that tell us what a healthy population, subject to minimal anthropogenic disturbance, should look like. Until recently, the only comparable trace element data for sea turtles came from stranded or free-ranging animals from other urban and industrially impacted regions of the world (39, 40, 41). Now, trace element baselines (for 20 elements) have been developed for green turtle blood using what is considered to be a healthy population from the Great Barrier Reef, a region that has historically been free from high levels of industrialisation (42). These baselines can be used to evaluate and monitor trace element trends in free-ranging green turtles, those in rehabilitation, or during mass stranding investigations. They also allow us to retrospectively evaluate trace element exposure from previously reported blood concentrations.
For example, by comparing the trace element concentrations reported in stranded green sea turtles (C. mydas) collected from Moreton Bay between 2006 and 2007 (38) to the recently developed baselines (42), the study showed that van de Merwe et al.’s concerns regarding high arsenic and cadmium were valid, being 10x and 4x the upper baseline limits, respectively. In addition, we also discovered that copper and particularly selenium were also elevated (1.4x and 6x, respectively) (Fig. 2). Due to a lack of relevant toxicological information for sea turtles (43), the overall health risks posed by elevated trace elements cannot be accurately predicted, particularly considering the complex mixtures of trace elements and other chemicals that sea turtles are exposed to in Moreton Bay (see also Case study 1).
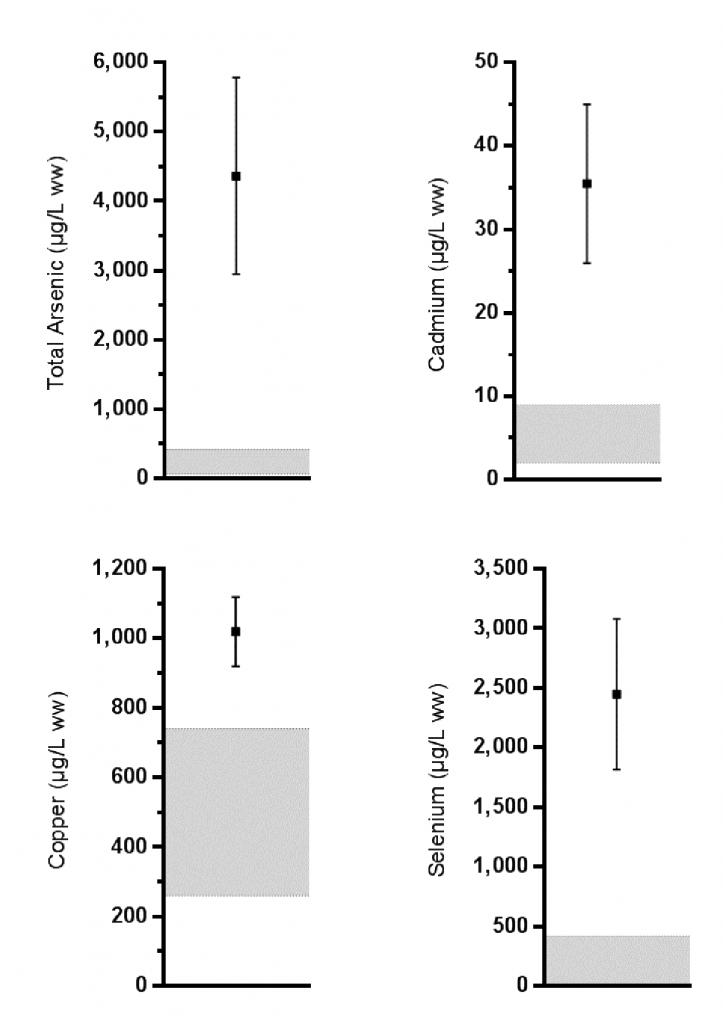
Trace element biomonitoring of populations of sea turtles and other marine megafauna that forage within Moreton Bay is important given the complex mixture of chemicals that the animals are exposed to and the dynamic physical and chemical fluctuations that occur. Additional baseline data, similar to those recently established for green turtles (Villa et al., 2017) is also required for other species, in order to better interpret biomonitoring data from free-ranging populations or as part of investigations into mass stranding. Finally, as recommended for POPs above, species-specific information on the toxic effects of trace element exposure in sea turtles and other species must be investigated to assess the impacts of trace elements accumulating in the Bay. Managers could then use this information when developing ecosystem indicators and local water quality guidelines.
Perfluoroalkyl substances
Perfluoroalkyl sulfonic acids (PFSAs) and perfluoroalkyl carboxylic acids (PFCAs) are an anthropogenic class of chemicals that have been used in numerous consumer products and industrial application since the 1950s. Sources of PFAAs include Teflon pans, outdoor clothing, firefighting foams, food packaging, carpets and pesticides. Some PFSAs and PFCAs bioaccumulate in biota and biomagnify along the food chain. They can present toxic effects, with some evidence suggesting that PFAAs could alter thyroid hormones levels in infants and pregnant women (44, 45). PFAAs are highly resistant to degradation, making them environmentally persistent. These chemicals have been detected worldwide, including remote areas such as Antarctica and lakes in the Himalayas (46). Their widespread distribution occurs through atmospheric transport, via surface water currents and via degradation of volatile precursors (47). Despite recent media attention (48), long-term monitoring data on PFAAs available within Australia are scant. Seven selected PFCAs and three selected PFSAs were detected in 100% of samples taken from the Parramatta River, the main tributary of Sydney Harbour, with a mean concentration ranging between 0.0002–0.014 µg/L (49), which is well within the current Recreational Water Quality Guidelines of 0.7 to 5.6 µg/L outlined by the Commonwealth Department of Health (50). Sampling of the Brisbane River system following major flooding in 2011 also found PFAAs in water from Somerset Dam to Moreton Bay (51). No manufacture of PFCAs or PFSAs has ever been recorded in Australia (52). Therefore, the presence of PFAAs in the Australian environment is due to the use and disposal of consumer articles (53) and the environmental degradation of other per and poly-fluorinated chemicals. The impact of PFAAs on Moreton Bay’s flora and fauna is not yet fully understood. In the meantime, the use and disposal of PFAA-generating consumer articles must be closely regulated.
Case study 3: Perfluoroalkyl substances and mass stingray stranding
PFSAs and PFCAs have been detected in the tissues of a wide range of aquatic species worldwide, particularly in liver tissue where they bioaccumulate (54). The stranding of 49 stingrays on North Stradbroke Island (Minjerribah) provided an opportunity to conduct tissue sampling (55). The concentrations of PFSAs and PFCAs were investigated in the liver tissues of six species of stingray (Aetobatus narinari, Dasyatis fluviorum, Himantura astra, Himantura toshi, Himantura uarnak, and Neotrygon kuhlii) with the aim of providing more information on the exposure level in Australia environment and assessing whether differences in concentrations could be explained as a function of the age/size of the individuals (55).
Perfluoroalkyl substances (PFAAs) were detectable in all liver samples, with total concentrations ranging from 1.2 to 152 ng·g−1 wet weight. The concentrations of total PFCAs and total PFSAs were significantly higher in blue-spotted rays (N. kuhlii) than in any other group of rays for all age/size categories (Fig. 3). PFAAs come in many forms and similar to previous studies on aquatic biota, perfluorooctane sulfonate (PFOS) was found to be the major contributor to total PFAAs in each population group.

In the case of rays, there is a positive relationship between the age and size, with larger rays being older (56). Interestingly there was a negative size-concentration relationship found for total PFAS levels in the blue-spotted rays (N. kuhlii), with larger rays having lower overall concentrations (55). This does appear to be counterintuitive, however, previous studies have shown that the uptake rates of PFAAs are faster and elimination rates slower in smaller fish (57). The rays were collected 20 months after the 2011 flood, when the Brisbane River delivered up to 17 kg of PFOS and 3.7 kg PFOA into Moreton Bay and showed a persistence of high concentration levels for many months post-flooding event (51). Therefore, the negative relationship between PFAAs concentration and size/age could be related to differing uptake kinetics of the chemicals as a response to an sudden increase in these substances 20 months prior, caused by the flooding of the Brisbane River. Hence, smaller rays take up the chemicals more quickly and eliminate them more slowly, leaving those rays with an overall larger body burden. This study highlighted the complexity around the uptake of PFAAs and the importance of including biological measurements such as size and age, in order to adequately study and understand the exposure of marine animals to these complex chemicals.
Marine debris
Marine debris is defined as any man-made product entering the marine environment, whether by accidental loss or damage, or wilful disposal. It is generally thought that about 20% of marine debris enters the water by being discarded or lost at sea, while about 80% originates from land-based sources (58). Land-based sources include littering and inadequate waste disposal facilities, with an estimated 33% of rubbish being lost from landfills into the environment every year (59). It is estimated that there is 150 billion tonnes of plastics in the world’s oceans as of 2017, with an estimated additional 4 to 12 million tonnes entering the world’s oceans every year (59). While the composition of this debris varies regionally and spatially, between 60 and 80% is plastic (60).
Global mass production of plastic commenced around 1950 at relatively low production levels of two million tonnes per year (59). However, it didn’t take long for these first plastic products to interact with marine life. The first scientific record of plastics being ingested was by seabirds in New Zealand during the late 1950s (61). This was less than 10 years after global mass production of plastics had started. Over the next couple of decades, plastic polymer production grew rapidly, with a compound annual growth rate of 8.4% a year (59). Today plastic debris production is estimated at 400 million tonnes per year and can be found in all of the oceans of the world (59, 62). Despite this rapid growth, the earliest recorded evidence of marine debris surveys in the Bay are from 1993 (63), by which time the global plastic production had increased to over 100 million tonnes per year (59).
In terms of threats to wildlife and potentially to human health, plastic debris is both persistent and pervasive (64). In different forms, it can be lethal through both ingestion and entanglement. Similarly, lost or discarded fishing gear such as nets, fishing line, and crab pots can entangle and drown marine life, while ingested hooks are lethal to both sea turtles and seabirds (65). A potential threat comes from microplastics, which are tiny pieces of plastic usually resulting from the deterioration of larger pieces of plastic or from clothing fibres (66). Microplastics can also adsorb chemicals from the environment and transfer them to the tissues of organisms that ingest them (8).
Case study 4: Marine debris, sea turtles and seabirds
The impacts to marine life from marine debris have been well catalogued (67), and include both lethal and sublethal impacts from entanglement and ingestion. Entangled animals can drown immediately, or can suffer from increased drag from entangled objects, impairing feeding and normal movement (68). Ingested objects can pierce the gut wall, causing septicaemia, or can block the gastrointestinal system (67). Recent research indicates that chemicals leached into body tissues from ingested plastics can cause hepatic stress, and even affect the endocrine system (8, 69).
A 2009 meta-analysis into the impacts of marine debris on wildlife in Australia found that 77 marine species had ingested or been entangled by plastic debris (70), but that number has certainly risen in the ensuing decade (e.g. (71)). In the Bay, the majority of scientific research on marine debris and wildlife to date has focused on sea turtles and seabirds (71-75). Whilst the population and ecosystem level effects of debris are more challenging to measure, there are several studies that have documented the effects of marine debris on a species level.
A survey of 115 sea turtles that died between 2006 and 2011 in Moreton Bay and nearby beaches found that 33% of all animals had ingested marine debris at the rate of anywhere between 1 and 329 pieces per turtle (73). Note, that these finding contrasts with Flint et al. (2010)(75) who found that only 4% (n=7) of necropsied sea turtles died from ingesting foreign bodies. However, the mean size of turtles in this study was much larger relative to Schuyler et al. (2012)(73). Interestingly, Schuyler et al. (2012)(73) noted that young, juvenile turtles, which had likely been foraging in pelagic offshore waters as opposed to inside of the Bay, were more likely to ingest debris (54.5%) than their elder counterparts (25%).
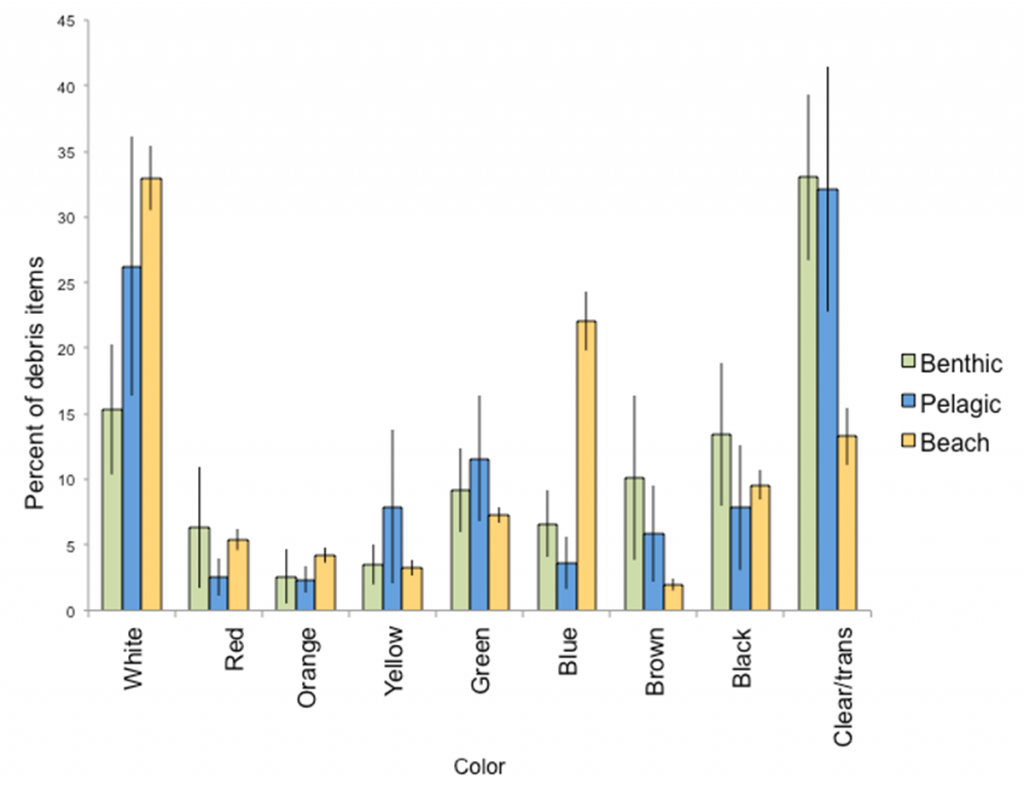
Schuyler et al. (2012)(73) also found differences between the type and colour of debris ingested by each group. Pelagic-feeding turtles preferred white, hard plastics, while the older, benthic-feeding turtles were more likely to eat soft, clear plastics (Fig. 4). The authors attributed this pattern to the ubiquity of white plastics in the environment and the relatively non-selective feeding style of post-hatchling turtles. In contrast, the preference of adult turtles may be due to the visual similarity of ingested plastic bags to a preferred prey taxa, jellyfish (73, 74). A quantitative analysis was made based on information obtained from the Queensland Government’s StrandNet and necropsies. This indicated that 14 pieces of marine debris were required to kill 50% of juvenile sea turtles investigated, many of which came from the Bay (76).
Similarly, a study of 139 short-tail shearwaters (Puffinus tenuirostris) stranded on the beaches of North Stradbroke Island found that 66% of all animals had ingested marine debris (72). Here again, juveniles were more likely to ingest debris (85%) than adults (63%). The birds appeared to select hard plastics, rubber and balloons. This selectivity may be due to a similarity in appearance (including position within the water column) between balloons and squid, a common prey item of shearwaters.
Although the animals were found on North Stradbroke Island, short-tail shearwaters (P. tenuirostris) migrate for thousands of kilometres, and the debris may have been ingested many kilometres from the Bay. However, this case study and that of the turtles indicate that wildlife frequenting the Bay and surrounds experience significant impacts from marine debris, whether the debris is local or regional in origin. It is critical to implement plans that reduce debris at its source across all levels of government, local, state and national.
Methods and bodies responsible for monitoring and cataloguing marine debris in Moreton Bay
Numerous organisations contribute to actions that deal with marine debris within the Bay by conducting community clean-ups and education. Three not-for-profit organisations—Healthy Land and Water, Reef Check Australia and Tangaroa Blue Foundation—are active leaders in monitoring and cataloguing debris with the goal to target debris sources in the Bay. These organisations collaborate with the Queensland Government’s Litter and Illegal Dumping Program to drive the adoption of best practices in waste management and education (77). Queensland’s Litter and Illegal Dumping Action Plan sets the agenda for managing the problem statewide. It is made up of a suite of programs that positively influence community attitudes and behaviours around waste disposal. Actions range from compliance and enforcement to community-based social-marketing campaigns, informed by rigorous research.
A better understanding of the sources of marine debris in the Bay is beginning to emerge from the data collected through standardised clean-ups conducted by Reef Check Australia, Healthy Land and Water Clean Up program, and Tangaroa Blue Foundation monitoring surveys and clean-ups (78). Reef Check Australia engages the community using a citizen science approach. They gather data that monitors and reports on reef health within the Moreton Bay Marine Park, including data on marine debris. The Healthy Land and Water Clean Up Program has run for over 16 years in south east Queensland and employs a crew to collect floating and bank-bound litter. Tangaroa Blue Foundation supports community clean-up events through an on-ground network of community volunteers and organisations, which contribute data on marine debris to the Australian Marine Debris Initiative database. It also provides educational resources and support programs, and collaborates with industry and government to create behavioural and legislative change at a state and national scale.
An important component of these programs is the systematic collection and analysis of data, which is used by the three organisations to educate and inform local communities and to drive local source reduction plans in collaboration with local and state governments. Within south east Queensland source reduction plans are being implemented to target specific waterway litter items. A source reduction plan serves the dual purpose of identifying regionally and temporally specific sources of litter and engaging the community, industry and local governments in a plan to reduce litter. The success of a source reduction plan can be evaluated using the data collected from community clean-up events. Through an iterative process of assessment, future reduction plans can evolve to address new issues as they arise.
In 2018, the Queensland Government banned single-use plastic bags (79) and mass balloon releases (80). During that same year, a container refund scheme for bottles, cans and cartons was also put into place (81). With ongoing beach/river clean-ups and data collection and analysis, source reduction plans can be measured for both success or failure, enabling them to be modified if needed, and providing case studies that can be used by communities across the country who may be trying to find similar solutions.
Conclusions
For all of the persistent pollutant groups discussed in this chapter, there is a recognised need to increase monitoring of these potentially toxic compounds, while simultaneously providing strategies to reduce their inputs into the Bay. These strategies require local, state and Australian government buy-in. Much progress has been made in the area of marine debris; however, many of the pollutants do not have coordinated monitoring programs, making it difficult to understand the impact of major weather events, such as flooding, into the Moreton Bay catchment.
The authors recognise that a coordinated monitoring program is not simple, as the number of industrial chemicals in use far exceeds the number of chemicals for which toxicological data are available (82). A proactive approach to developing baselines is complicated as it is difficult to predict what will be an issue and it is impractical to make baselines for all potentially toxic chemicals in the short term. One way to address this issue is to generate an environmental specimen bank that contains tissue samples that can be accessed by researchers and government agencies to go ‘back in time’ to make comparisons.
Queensland lacks a well-established or coordinated system for necropsies/sampling of stranded animals. It is highly recommended that the Queensland Government, in conjunction with a relevant tertiary institution, such as the University of Queensland’s ‘Queensland Alliance for Environmental Health Science’, look towards establishing a well- maintained and well-funded tissue bank, such as the National Oceanic and Atmospheric Administration’s marine mammal tissue bank (83) or the South Australian Museum tissue bank (84). It is also recommended that researchers consider the practical applications of their work by working with, or developing tools for, frontline conservation and monitoring agencies. It is time to move on from monitoring problems and actively concentrate our energy on solution-based approaches.
References
- History B. 2017. Brisbane history – a history of old Brisbane to 1900. [Accessed: 25/1/2017 2017]. Available from: http://www.brisbanehistory.com/.
- Council BC. 2017. Brisbane key economic facts. [Accessed: 25/1/2017 2017]. Available from: https://www.brisbane.qld.gov.au/about-council/governance-strategy/business-brisbane/growing-brisbanes-economy/brisbanes-key-economic-facts
- Naito W, Jin HC, Kang YS, Yamamuro M, Masunaga S, Nakanishi J. 2003. Dynamics of PCDDS/DFS and coplanar-PCBS in an aquatic food chain of Tokyo Bay. Chemosphere. 53(4):347-362. http://dx.doi.org/10.1016/s0045-6535(03)00046-8
- Lallas PL. 2001. The Stockholm Convention on persistent organic pollutants. American Journal of International Law. 95(3):692-708. http://dx.doi.org/10.2307/2668517
- Jan MR, Shah J, Khawaja MA, Gul K. 2009. DDT residue in soil and water in and around abandoned DDT manufacturing factory. Environmental Monitoring and Assessment. 155(1-4):31-38. http://dx.doi.org/10.1007/s10661-008-0415-2
- Niimi AJ. 1996. Evaluation of PCBS and PCDD/FS retention by aquatic organisms. Science of The Total Environment. 192(2):123-150. http://dx.doi.org/10.1016/s0048-9697(96)05306-5
- Skene SA, Dewhurst IC, Greenberg M. 1989. Polychlorinated dibenzo-p-dioxins and polychlorinated dibenzofurans: The risks to human health. A review. Human Toxicology. 8(3):173-203. http://dx.doi.org/10.1177/096032718900800301
- Rochman CM, Hoh E, Kurobe T, Teh SJ. 2013. Ingested plastic transfers hazardous chemicals to fish and induces hepatic stress. Scientific reports. 3
- Rochman CM, Kurobe T, Flores I, Teh SJ. 2014. Early warning signs of endocrine disruption in adult fish from the ingestion of polyethylene with and without sorbed chemical pollutants from the marine environment. Science of The Total Environment. 493:656-661
- Gaus C, Päpke O, Dennison N, Haynes D, Shaw GR, Connell DW, Mueller JF. 2001. Evidence for the presence of a widespread PCDD source in coastal sediments and soils from Queensland, Australia. Chemosphere. 43(4-7):549-558
- Matthews V, Päpke O, Burns D, Gaus C. 2011. Contaminant dietary exposure assessment for a coastal community in Moreton Bay, Queensland: Persistent organic pollutants in local seafood. Proceedings of the Royal Society of Queensland. 117:455-466
- Matthews V, Päpke O, Gaus C. 2008. PCDD/FS and PCBs in seafood species from Moreton Bay, Queensland, Australia. Marine Pollution Bulletin. 57:392-402
- Hermanussen S. 2009. Distribution and fate of persistent organic pollutants in nearshore marine turtle habitats of Queensland, Australia. PhD Thesis, The University of Queensland
- Weijs L, Vijayasarathy S, Villa CA, Neugebauer F, Meager JJ, Gaus C. 2016. Screening of organic and metal contaminants in Australian humpback dolphins (Sousa sahulensis) inhabiting an urbanised emBayment. Chemosphere. 151:253-262. http://dx.doi.org/10.1016/j.chemosphere.2016.02.082
- Ageing AGDoHa. 2004. Human health risk assessment of dioxins in Australia. Australian Government Department of the Environment and Heritage. Canberra
- Deutch B, Pedersen HS, Hansen JC. 2004. Dietary composition in Greenland 2000, plasma fatty acids and persistent organic pollutants. Science of The Total Environment. 331(1-3):117-188
- Van Oostdam J, Donaldson SG, Feeley M, Arnold D, Ayotte P, Bondy G, Chan L, Dewaily E, Furgal CM, Kuhnlein H. 2005. Human health implications of environmental contaminants in arctic Canada: A review. Science of The Total Environment. 351-352:165-246
- Carter P, Durbidge E, Cooke-Bramley J. 1994. Historic North Stradbroke Island. North Stradbroke Island Historical Museum Association, Dunwich, QLD
- Veronica M. 2009. Contaminant dietary exposure assessment for a coastal subpopulation in Queensland, Australia. PhD Thesis, The University of Queensland, School of Medicine.
- Haynes D, Johnson JE. 2000. Organochlorine, heavy metal and polyaromatic hydrocarbon pollutant concentrations in the Great Barrier Reef (Australia) environment: A review. Marine Pollution Bulletin. 41(7-12):267-278. http://dx.doi.org/10.1016/S0025-326x(00)00134-X
- Brady JP, Ayoko GA, Martens WN, Goonetilleke A. 2014. Temporal trends and bioavailability assessment of heavy metals in the sediments of Deception Bay, Queensland, Australia. Marine Pollution Bulletin. 89(1-2):464-472. http://dx.doi.org/10.1016/j.marpolbul.2014.09.030
- Dunn RJK, Teasdale PR, Warnken J, Jordan MA, Arthur JM. 2007. Evaluation of the in situ, time-integrated DGT technique by monitoring changes in heavy metal concentrations in estuarine waters. Environmental Pollution. 148(1):213-220. http://dx.doi.org/10.1016/j.envpol.2006.10.027
- Coates-Marnane J, Olley J, Burton J, Grinham A. 2016. The impact of a high magnitude flood on metal pollution in a shallow subtropical estuarine emBayment. Sci Total Environ. 569-570:716-731. http://dx.doi.org/10.1016/j.scitotenv.2016.06.193
- Aggett P, Nordberg GF, Nordberg M. 2015. Essential metals: Assessing risks from deficiency and toxicity. In: Aggett P, Nordberg GF, Nordberg M (Eds). Handbook on the toxicology of metals (fourth edition). Academic Press, San Diego. p. 283-284. http://dx.doi.org/10.1016/B978-0-444-59453-2.00014-7
- Luoma SN, Rainbow PS. 2005. Why is metal bioaccumulation so variable? Biodynamics as a unifying concept. Environmental Science & Technology. 39(7):1921-1931. http://dx.doi.org/10.1021/es048947e
- Goonetilleke A, Egodawatta P, Kitchen B. 2009. Evaluation of pollutant build-up and wash-off from selected land uses at the Port of Brisbane, Australia. Marine Pollution Bulletin. 58(2):213-221. http://dx.doi.org/10.1016/j.marpolbul.2008.09.025
- Morelli G, Gasparon M. 2014. Metal contamination of estuarine intertidal sediments of Moreton Bay, Australia. Mar Pollut Bull. 89(1-2):435-443. http://dx.doi.org/10.1016/j.marpolbul.2014.10.002
- Brady JP, Ayoko GA, Martens WN, Goonetilleke A. 2014. Enrichment, distribution and sources of heavy metals in the sediments of Deception Bay, Queensland, Australia. Marine Pollution Bulletin. 81(1):248-255. http://dx.doi.org/10.1016/j.marpolbul.2014.01.031
- Cox ME, Preda M. 2005. Trace metal distribution within marine and estuarine sediments of western Moreton Bay, Queensland, Australia: Relation to land use and setting. Geographical Research. 43(2):173-193. http://dx.doi.org/10.1111/j.1745-5871.2005.00312.x
- Prange JA, Dennison WC. 2000. Physiological responses of five seagrass species to trace metals. Marine Pollution Bulletin. 41(7-12):327-336. http://dx.doi.org/10.1016/S0025-326x(00)00126-0
- Ansmann IC, Lanyon JM, Seddon JM, Parra GJ. 2015. Habitat and resource partitioning among indo-pacific bottlenose dolphins in Moreton Bay, Australia. Marine Mammal Science. 31(1):211-230. http://dx.doi.org/10.1111/mms.12153
- Richards RG, Chaloupka M. 2008. Does oyster size matter for modelling trace metal bioaccumulation? Science of The Total Environment. 389(2–3):539-544. http://dx.doi.org/10.1016/j.scitotenv.2007.08.060
- Clark MW. 1998. Management implications of metal transfer pathways from a refuse tip to mangrove sediments. Science of The Total Environment. 222(1–2):17-34. http://dx.doi.org/10.1016/S0048-9697(98)00283-6
- Morelli G, Gasparon M, Fierro D, Hu WP, Zawadzki A. 2012. Historical trends in trace metal and sediment accumulation in intertidal sediments of Moreton Bay, southeast Queensland, Australia. Chemical Geology. 300:152-164. http://dx.doi.org/10.1016/j.chemgeo.2012.01.023
- Unit WQaEHP. 2010. Environmental protection (water) policy 2009 – Moreton Bay environmental values and water quality objectives. Queensland Government. Brisbane, QLD. Available from: https://environment.des.qld.gov.au/water/policy/pdf/documents/moreton-Bay-ev-2010.pdf
- Division EPaP. 2018. Guideline: Environmental protection (water) policy 2009 – deciding aquatic ecosystem indicators and local water quality guidelines. The Queensland Government, Science DoEa. Brisbane, QLD. Available from: https://environment.des.qld.gov.au/water/pdf/deriving-local-water-quality-guidelines.pdf. December
- Gordon AN, Pople AR, Ng J. 1998. Trace metal concentrations in livers and kidneys of sea turtles from south-eastern Queensland, Australia. Marine and Freshwater Research. 49(5):409-414. http://dx.doi.org/10.1071/mf97266
- van de Merwe JP, Hodge M, Olszowy HA, Whittier JM, Lee SY. 2010. Using blood samples to estimate persistent organic pollutants and metals in green sea turtles (Chelonia mydas). Marine Pollution Bulletin 60(4):579-588. http://dx.doi.org/10.1016/j.marpolbul.2009.11.006
- Gaus C, Grant S, Jin NL, Goot K, Chen L, Villa CA, Neugebauer F, Qi L, Limpus C. 2012. Investigations of contaminant levels in green turtles from Gladstone. The University of Queensland, Australia. Report No.: Final Report. National Research Centre for Environmental Toxicology. Available from: http://gladstoneconservationcouncil.com.au/web/wp-content/uploads/2013/04/Entox-Report-Investigation-of-contaiminanr-Levels-in-Green-Turtles-from-Gladstone.pdf
- Storelli MM, Marcotrigiano GO. 2003. Heavy metal residues in tissues of marine turtles. Marine Pollution Bulletin. 46(4):397-400. 10.1016/S0025-326X(02)00230-8
- Flint M, Morton JM, Limpus CJ, Patterson-Kane JC, Mills PC. 2010. Reference intervals for plasma biochemical and hematologic measures in loggerhead sea turtles (Caretta caretta) from Moreton Bay, Australia. Journal of Wildlife Diseases. 46(3):731-741. http://dx.doi.org/10.7589/0090-3558-46.3.731
- Villa CA, Flint M, Bell I, Hof C, Limpus CJ, Gaus C. 2017. Trace element reference intervals in the blood of healthy green sea turtles to evaluate exposure of coastal populations. Environmental Pollution. 220(Pt B):1465-1476. http://dx.doi.org/10.1016/j.envpol.2016.10.085
- Finlayson KA, Leusch FD, van de Merwe JP. 2016. The current state and future directions of marine turtle toxicology research. Environment International. 94:113-123. http://dx.doi.org/10.1016/j.envint.2016.05.013
- Lau C. 2015. Perfluorinated compounds: An overview. In: DeWitt JC (Ed.). Toxicological effects of perfluoroalkyl and polyfluoroalkyl substances, molecular and integrative toxicology. Springer International Publishing. p. 1-21. http://dx.doi.org/10.1007/978-3-319-15518-0_1
- Wang Y, Rogan WJ, Chen PC, Lien G-W, Chen HY, Tseng YC, Longnecker MP, Wang SL. 2014. Association between maternal serum perfluoralkyl substances during pregnancy and maternal and cord thyroid hormones: Taiwan maternal and cohort study. Environmental Health Perspectives. 122(529-534)
- Ahrens L. 2011. Polyfluoroalkyl compounds in the aquatic environment: A review of their occurrence and fate. Journal of Environmental Monitoring. 13:20-31. http://dx.doi.org/10.1039/C0EM00373E
- Buck RC, Franklin J, Berger U, Conder JM, Cousins IT, de Voogt P, Jenson AA, Kannan K, Mabury SA, van Leeuwen SP. 2011. Perfluoroalkyl and polyfluoroalkyl substances in the environment: Terminology, classification, and origins. Integrated Environmental Assessment Management. 7:513-541. http://dx.doi.org/10.1002/ieam.258
- Fellner C, Begley P. 2018. Toxic secrets: Where the stites with PFAS contamination are near you. The Sydney Morning Herald
- Thompson J, Roach A, Eaglesham G, Bartkow ME, Edge K, Mueller JF. 2011. Perfluorinated alkyl acids in water, sediment and wildlife from Sydney Harbour and surroundings. Marine Pollution Bulletin. 62:2869-2875. http://dx.doi.org/10.1016/j.marpolbul.2011.09.002
- Health E. 2017. PFOS and PFOA. [Accessed: February 18 2019]. Available from: https://www.health.nsw.gov.au/environment/factsheets/Pages/pfos.aspx
- Gallen C, Baduel C, Lai FY, Thompson K, Thompson J, Warne M, Mueller JF. 2014. Spatio-temporal assessment of perfluorinated compounds in the Brisbane river system, Australia: Impact of a major flood event. Marine Pollution Bulletin. 85:597-605
- NICNAS. 2013. Derivatives and chemicals on which they are based: Alert factsheet. In: Scheme NICNaA, editor. National Industrial Chemicals Notification and Assessment Scheme. Sydney, Australia
- Gallen C, Drage D, Kaserzon S, Baduel C, Gallen C, Banks A, Broomhall S, Mueller JF. 2016. Occurrence and distribution of brominated flame retardants and perfluoroalkyl substances in Australian landfill leachate and biosolids. Journal of Hazardous Materials. 312:55-64. http://dx.doi.org/10.1016/j.jhazmat.2016.03.031
- Houde M, De Silva AO, Muir DCG, Letcher RJ. 2011. Monitoring of perfluorinated compounds in aquatic biota: An updated review: PFCS in aquatic biota. Environment Science and Technology. 45:7962-7973. http://dx.doi.org/10.1021/es104326w
- Baduel C, Lai FY, Townsend KA, Mueller JF. 2014. Size and age–concentration relationships for perfluoroalkyl substances in stingray livers from eastern Australia. Science of The Total Environment. 496:523-530. http://dx.doi.org/10.1016/j.scitotenv.2014.07.010
- Yigin CC, Ismen A. 2012. Age, growth and reproduction of the common stingray, Dasyatis pastinaca from the north Aegean Sea. Marine Biology Research. 8(7):644-653. http://dx.doi.org/10.1080/17451000.2012.659667
- Arnot JA, Gobas F. 2006. A review of bioconcentration factor (BCF) and bioaccumulation factor (BAF) assessments for organic chemicals in aquatic organisms. Environmental Reviews. 14(4):257-297. http://dx.doi.org/10.1139/a06-005
- Faris J, Hart KM. Seas of debris: A summary of the third international conference on marine debris. Third International Conference on Marine Debris; 1994; Raleigh, North Carolina: North Carolina Sea Grant College Program
- Geyer R, Jambeck JR, Law KL. 2017. Production, use, and fate of all plastics ever made. Science Advances. 3(7), e1700782. http://dx.doi.org/10.1126/sciadv.1700782
- Derraik JGB. 2002. The pollution of the marine environment by plastic debris: A review. Marine Pollution Bulletin. 44(9):842-852
- Harper PC, Fowler JA. 1987. Plastic pellets in New Zealand storm-killed prions (Pachyptila spp.). Notornis. 34:65-70
- Barnes DKA, Galgani F, Thompson RC, Barlaz M. 2009. Accumulation and fragmentation of plastic debris in global environments. Philosophical Transactions of the Royal Society B-Biological Sciences. 364(1526):1985-1998. http://dx.doi.org/10.1098/rstb.2008.0205
- O’Callaghan P. 1993. Sources of coastal shoreline litter near three Australian cities. Victorian Institute of Marine Sciences. Queenscliff, Victoria
- Ogata Y, Takada H, Mizukawa K, Hirai H, Iwasa S, Endo S, Mato Y, Saha M, Okuda K, Nakashima A, Murakami M, Zurcher N, Booyatumanondo R, Zakaria MP, Dung LQ, Gordon M, Miguez C, Suzuki S, Moore C, Karapanagioti HK, Weerts S, McClurg T, Burres E, Smith W, Van Velkenburg M, Lang JS, Lang RC, Laursen D, Danner B, Stewardson N, Thompson RC. 2009. International pellet watch: Global monitoring of persistent organic pollutants (POPS) in coastal waters. 1. Initial phase data on PCBs, DDTs, and HCHS. Marine Pollution Bulletin. 58(10):1437-1446. http://dx.doi.org/10.1016/j.marpolbul.2009.06.014
- McPhee DP, Leadbitter D, Skilleter G. 2002. Swallowing the bait: Is recreational fishing in Australia ecologically sustainable? Pacific Conservation Biology. 8:40-51
- Halstead JE, Smith JA, Carter EA, Lay PA, Johnston EL. 2018. Assessment tools for microplastics and natural fibres ingested by fish in an urbanised estuary. Environmental Pollution. 234:552-561. http://dx.doi.org/10.1016/j.envpol.2017.11.085
- Nelms SE, Duncan EM, Broderick AC, Galloway TS, Godfrey MH, Hamann M, Lindeque PK, Godley BJ. 2015. Plastic and marine turtles: A review and call for research. ICES Journal of Marine Science. 73(2):165-181. http://dx.doi.org/10.1093/icesjms/fsv165
- Laist D. 1997. Impacts of marine debris: Entanglement of marine life in marine debris including a comprehensive list of species with entanglement and ingestion records. In: Coe JM, Rogers DB (Eds). Marine debris: Sources, impacts, and solutions. Springer, New York. p. 99-139
- Rochman CM, Kurobe T, Flores I, Teh SJ. 2014. Early warning signs of endocrine disruption in adult fish from the ingestion of polyethylene with and without sorbed chemical pollutants from the marine environment. Science of the Total Environment. 493:656-661
- Ceccarelli DM. 2009. Impacts of plastic debris on Australian marine wildlife. Canberra
- Roman L, Schuyler QA, Hardesty BD, Townsend KA. 2016. Anthropogenic debris ingestion by avifauna in eastern Australia. PLoS ONE. 11(8). http://dx.doi.org/10.1371/journal.pone.0158343
- Acampora H, Schuyler QA, Townsend KA, Hardesty BD. 2014. Comparing plastic ingestion in juvenile and adult stranded short-tailed shearwaters (Puffinus tenuirostris) in eastern Australia. Marine Pollution Bulletin. 78(1-2):63-68. http://dx.doi.org/10.1016/j.marpolbul.2013.11.009
- Schuyler Q, Hardesty BD, Wilcox C, Townsend K. 2012. To eat or not to eat? Debris selectivity by marine turtles. PLoS ONE. 7(7). e40884 10.1371/journal.pone.0040884
- Schuyler QA, Wilcox C, Townsend K, Hardesty BD, Marshall NJ. 2014. Mistaken identity? Visual similarities of marine debris to natural prey items of sea turtles. BMC Ecology. 14. http://dx.doi.org/10.1186/1472-6785-14-14
- Flint M, Patterson-Kane JC, Limpus CJ, Mills PC. 2010. Health surveillance of stranded green turtles in southern Queensland, Australia (2006-2009): An epidemiological analysis of causes of disease and mortality. Ecohealth. 7(1):135-145. http://dx.doi.org/10.1007/s10393-010-0300-7
- Wilcox C, Puckridge M, Schuyler QA, Townsend K, Hardesty BD. 2018. A quantitative analysis linking sea turtle mortality and plastic debris ingestion. Scientific Reports. 8: 12536. http://dx.doi.org/10.1038/s41598-018-30038-z
- Government Q. 2013. Queensland’s litter and illegal dumping action plan. State of Queensland, Protection DoEaH. Brisbane, Queensland. Available from: https://www.qld.gov.au/environment/assets/documents/pollution/management/waste/qld-litter-illegal-dump-action-plan.pdf. October 2013
- Taylor H. 2016. Tangaroa blue foundation. [Accessed: Jan 21, 2017 2017]. Available from: www.tangaroablue.org
- Government Q. 2018. Plastic bag ban. [Accessed: March 30 2018]. Available from: https://www.qld.gov.au/environment/pollution/management/waste/plastic-bags
- Governement Q. 2017. Releasing balloons. [Accessed: March 30 2018]. Available from: https://www.qld.gov.au/environment/pollution/management/waste/balloons
- Government Q. 2018. Container refund scheme. [Accessed: March 30 2018]. Available from: https://www.qld.gov.au/environment/pollution/management/waste/container-refund
- Tang JYM, Busetti F, Charrois JWA, Escher BI. 2014. Which chemicals drive biological effects in wastewater and recycled water? Water Research. 60:289-299. http://dx.doi.org/10.1016/j.watres.2014.04.043
- Fisheries N. 2017. National marine mammal tissue bank (NMMTB). [Accessed: 2 April 2108]. Available from: http://www.nmfs.noaa.gov/pr/health/tissue/
- Wheaton L. 2018. Australian biological tissue collection. [Accessed: 2 April 2018]. Available from: sa.gov.au/collections/biological-sciences/biological-tissues
- Aguilar A, Borrell A. 1994. Abnormally high polychlorinated biphenyl levels in striped dolphins (Stenella coeruleoalba) affected by the 1990–1992 Mediterranean epizootic. Science of The Total Environment. 154(2–3):237-247. http://dx.doi.org/10.1016/0048-9697(94)90091-4
- DeLong RL, Gilmartin WG, Simpson JG. 1973. Premature births in California sea lions: Association with high organochlorine pollutant residue levels. Science. 181(4105):1168-1170. http://dx.doi.org/10.1126/science.181.4105.1168
- Helle E, Olsson M, Jensen S. 1976. PCB levels correlated with pathological changes in seal uteri. Ambio. 5(5/6):261-262. http://dx.doi.org/10.2307/4312230
- Jepson PD, Bennett PM, Deaville R, Allchin CR, Baker JR, Law RJ. 2005. Relationships between polychlorinated biphenyls and health status in harbor porpoises (Phocoena phocoena) stranded in the United Kingdom. Environmental Toxicology and Chemistry. 24(1):238-248. http://dx.doi.org/10.1897/03-663.1
- de Swart RL, Ross PS, Vos JG, Osterhaus AD. 1996. Impaired immunity in harbour seals (Phoca vitulina) exposed to bioaccumulated environmental contaminants: Review of a long-term feeding study. Environmental Health Perspectives. 104(Suppl 4):823-828
