-
Previous paper
Managing for the multiple uses and values of Moreton Bay and its catchments
Helen Ross1, David Rissik 2,3, Natalie Jones1, Katherine Witt4, Breanna Pinner5, Sylvie Shaw5 -
This paper
Performance of marine reserves for fish and associated ecological functions in the Moreton Bay Marine Park
Ben L. Gilby1, Andrew D. Olds1, David Rissik2,3, Christopher J. Henderson1,4, Rod M. Connolly4, Tim Stevens4 and Thomas A. Schlacher1 -
Next paper
Changes in fish and crab abundance in response to the Moreton Bay Marine Park rezoning
Michael Haywood1, Richard Pillans1, Russ Babcock1, Emma Lawrence2, Ross Darnell2, Charis Burridge2, Darren Dennis1, Anthea Donovan1, Sue Cheers1, Robert Pendrey1, and Quinton Dell1
Performance of marine reserves for fish and associated ecological functions in the Moreton Bay Marine Park
Authors
Ben L. Gilby1, Andrew D. Olds1, David Rissik2,3, Christopher J. Henderson1,4, Rod M. Connolly4, Tim Stevens4 and Thomas A. Schlacher1Author affiliations
- School of Science and Engineering, University of the Sunshine Coast, Maroochydore DC Qld, 4558, Australia;
- BMT Eastern Australia Pty. Ltd, Brisbane, Qld 4000, Australia
- Australian Rivers Institute, Griffith University, Nathan, QLD 4111, Australia
- Australian Rivers Institute – Coasts and Estuaries, Griffith University, Gold Coast Qld, 4222, Australia
Corresponding author
bgilby@usc.edu.auORCID
Ben Gilby: https://orcid.org/0000-0001-8642-9411
Andrew Olds: https://orcid.org/0000-0002-8027-3599
David Rissik: https://orcid.org/0000-0002-1976-5324
Christopher Henderson: https://orcid.org/0000-0001-9397-8064
Rod Connolly: https://orcid.org/0000-0001-6223-1291
Tim Stevens: https://orcid.org/0000-0002-0876-5169
Thomas Schlacher: https://orcid.org/0000-0003-2184-9217
Book
Performance of marine reserves for fish and associated ecological functions in the Moreton Bay Marine Park
Chapter
Research Paper Title
Performance of marine reserves for fish and associated ecological functions in the Moreton Bay Marine Park
Cite this paper as:
Gilby B, Olds A, Rissik D, Henderson C, Connolly R, Stevens T, Schlacher T. 2019. Performance of marine reserves for fish and associated ecological functions in the Moreton Bay Marine Park. In Tibbetts, I.R., Rothlisberg, P.C., Neil, D.T., Homburg, T.A., Brewer, D.T., & Arthington, A.H. (Editors). Moreton Bay Quandamooka & Catchment: Past, present, and future. The Moreton Bay Foundation. Brisbane, Australia. Available from: https://moretonbayfoundation.org/
DOI
10.6084/m9.figshare.8085731
ISBN
978-0-6486690-0-5
Abstract
No-take marine reserves in Moreton Bay were established to conserve and restore the structure and function of marine ecosystems and ensure sustainable social-ecological systems (Ross et al. 2019, this volume). Here, we review published literature to determine our current understanding of how no-take marine reserves (i.e. green zones) benefit fish, and shape ecological functions in numerous ecosystems within the Moreton Bay Marine Park. Over the past decade, 16 peer-reviewed studies have examined ‘reserve effects’ for fish and associated ecological processes in Moreton Bay; this work was mostly conducted in the central part of the Bay in coral reefs and seagrass meadows. Most studies showed enhanced fish abundance, diversity, or both, inside reserves and increases in the levels of functions performed by fish (e.g. greater grazing rates). The degree to which reserves enhance the abundance of fish and their functions was contingent on two key conditions. Reserves that were better connected (i.e. those in proximity to complementary habitat types and situated in a more complex seascape) and those located in clearer water typically perform better and may also be more resilient. Therefore, these two factors must be strongly considered during future deliberations about expanding or modifying reserves in Moreton Bay. We identify a number of information gaps that are likely to impede improvement to the current network of no-take reserves, namely, research on sandy and muddy bottoms, ocean beaches, fishing outside of reserves, and pivotal ecological functions other than herbivory. Reserve design and future rezoning can take advantage of the considerable body of evidence gathered on factors governing reserve performance for fishes, but reserve effects need to be examined for the numerous habitats that have been overlooked. How reserves shape a broader range of functions, productivity, habitat and ecological resilience needs to be investigated.
Keywords: conservation planning, coral reef, disturbance, fisheries, habitat, offshore, seagrass, spatial ecology, water quality
Introduction
No-take marine reserves (i.e. green zones) are widely used to conserve habitats and species in the face of human threats, particularly fishing (1). Many studies have assessed the efficacy of no-take reserves for fishes over the past two decades. Reviews of the global literature show that strategically placed and well-enforced marine reserves can benefit populations and assemblages of fishes, especially species that are heavily harvested (2, 3). Importantly, marine reserves can in some instances promote the recovery of over-fished apex predators and herbivores towards their pre-fished biomass, thereby reversing trophic cascades (4, 5), restoring ecological functions (e.g. herbivory, predation, recruitment), and the condition of some benthic ecosystems (3, 4, 6).
Historically, research on reserves mainly asked whether they enhanced the biomass of fished species or restored food webs (7, 8). Recent work has expanded in scope, asking inter alia: (i) whether reserves can benefit surrounding fished areas (i.e.spillover)(9,10), (ii) how effective reserves are compared with other management interventions (11), and (iii) whether and how reserves can increase ecological resilience (i.e. the capacity of ecosystems to resist, or recover from, disturbance). It is also increasingly acknowledged that reserve effects extend beyond fishes and other mobile animals (12). The key ecological functions of herbivory, predation and scavenging can be significantly modified in intensity and spatial extent within reserves, and might improve the capacity of ecosystems to withstand disturbance (13-15). For example, in Kenyan marine reserves, the recovery of keystone predators reduced the abundance of their sea urchin prey (16), and promoted recovery of calcifying algae (17). These are critical advances in our understanding of reserve effectiveness for ecosystems (18, 19).
Research on reserves within the Moreton Bay Marine Park, has contributed measurably to the global literature on the topic (3, 20, 21), and in the past decade, has helped to progress our understanding of how reserves influence fish communities (22, 23), ecological functions (24, 25), and the structure and resilience of ecosystems (26-28). The quantity and quality of published outputs from Moreton Bay on these topics is comparable to the Caribbean (4), Kenya (29) and Tasmania (30), which are widely regarded as prime research areas on this topic (3). In this study, we review the peer-reviewed, published literature for studies that quantify how no-take reserves influence fish assemblages and ecological functions performed by fishes in Moreton Bay. We identify the habitats where these studies were focused, the types of factors the studies considered in their analysis that might modify overall effects, and whether the study concluded that the reserves were overall ‘effective’ for their target groups, species, or functions within Moreton Bay.
Summary of research on reserve effectiveness
Individual species and specific habitats in Moreton Bay have been provided various forms of protection since the late 1800s, and the first marine park with formalised zoning was implemented in Moreton Bay in 1993 (Ross et al. 2019, this volume). Despite this long history of protection, research into conservation performance was not published until 2005 (31), and did not focus on fish until 2007 (32). Since then, 16 studies have examined reserve effectiveness for fishes and/or ecological functions in Moreton Bay (Table 1). Each of these studies sampled multiple no-take reserves and compared them with multiple reference sites that were open to fishing. Only one article charted how assemblages in reserves changed relative to their condition prior to reserve establishment (33).
Table 1. Summary of studies on reserve performance in Moreton Bay, grouped by ecosystem type. UVC: underwater visual census, BRUVS: baited remote underwater video stations. ‘Reserves effective?’ is an assessment of whether the study in question shows positive effects of marine reserves on either assemblages or ecological functions (Y, effective; Y*, somewhat effective; N, not effective).
| Reference | Survey method | Focal species/group | Focal function | External factors | Reserves effective? |
Coral reefs |
|||||
| Olds et al. 2012 (27) | UVC | Fish assemblage | Herbivory | Seascape | Y* |
| Olds et al. 2012 (22) | UVC | Fish assemblage | – | Seascape | Y* |
| Olds et al. 2014 (28) | UVC | Fish assemblage | Herbivory | Water quality, seascape | Y* |
| Gilby et al. 2015 (11) | Benthic quadrats, algae deployments | Benthic macroalgae | Herbivory | Water quality | N |
| Yabsley et al. 2016 (25) | UVC, algae deployments | Fish assemblage (herbivores) | Herbivory (grazing and browsing) | Seascape | Y* |
| Gilby et al. 2016 (23) | Benthic quadrats, BRUVS | Fish assemblage | Predation | Water quality, seascape | N |
| Gilby et al. 2016 (24) | BRUVS | Fish assemblage (herbivores) | Herbivory (browsing) | – | N |
| Gilby et al. 2016 (49) | Benthic quadrats/BRUVS | Fish assemblage | Predation, herbivory (grazing and browsing) | Water quality, seascape | N |
Seagrass |
|||||
| Pillans et al. 2007 (32) | Seine net | Nekton | – | – | Y |
| Henderson et al. 2017
(35) |
BRUVS | Fish assemblage (harvested) | – | Water quality, Seascape | Y* |
| Henderson et al. 2017 (26) | Algae deployments, BRUVS | Fish assemblage (harvested) | Herbivory (browsing) | Seascape positioning | N |
| Henderson et al. 2017 (42) | Acoustic telemetry | Giant shovelnose ray (Glaucostegus typus) | – | Seascape positioning | N |
Mangroves |
|||||
| Olds et al. 2012 (22) | UVC | Fish assemblage | – | Seascape | Y* |
Estuaries |
|||||
| Pillans et al. 2007 (32) | Seine net | Nekton | – | – | Y |
| Olds et al. 2017 (56) | BRUVS | Fish assemblage | Scavenging | Seascape positioning, urbanisation | N |
| Gilby et al. 2017 (39) | BRUVS | Fish assemblage | – | Seascape positioning, urbanisation, water quality | N |
Offshore rocky reefs |
|||||
| Terres et al. 2015 (33) | BRUVS | Pink snapper | – | Habitat type and positioning | Y |
| Unconsolidated soft sediments | |||||
| Ortodossi et al. 2018 (40) | BRUVS | Fish assemblage | – | Seascape positioning | Y* |
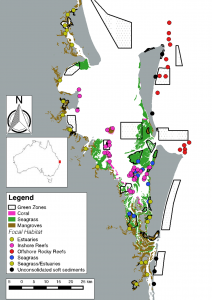
In terms of ecosystem coverage, studies can be grouped broadly into those focusing on one or two of the six main ecosystem types (Figs. 1, 2). Coral reefs were the most researched ecosystem in Moreton Bay, with eight studies overall, followed by seagrass meadows (four studies). Outside of these ecosystems, estuaries (three studies), mangroves (one study), deep reefs (>20m water depths) in areas offshore of bordering sand islands (one study), and unconsolidated soft sediments in the nearshore surf zones of sandy beaches (one study) have also been assessed (Figs. 1, 2). For the purposes of this study, ‘estuaries’ were defined as the tidal mouth of a creek or river, where the tide meets the stream (which can often include mangroves), whereas mangroves were defined as any areas outside of this definition which surveyed ‘mangrove’ ecosystems. Most research (11 of 16 included studies) has been conducted in the heterogeneous seascapes of central Moreton Bay (Fig. 1). Here, the close proximity of mangroves, seagrasses and corals allows for research on both ecosystem-specific responses to marine reserves, as well as determining how connectivity between ecosystems modifies reserve effects.
Thematically, studies have focused on either one or a combination of four main topics: (i) fish communities (fish abundance and assemblage composition), (ii) ecological functions (herbivory, scavenging), (iii) benthic ecosystems (seagrasses and corals), and (iv) reserve effects in the context of disturbance and complementary management interventions (water-quality effects, disturbance from floods, spatial effects of seascape connectivity on reserve performance) (Fig. 2, Table 1). A sizeable fraction (40%) of studies examined various facets of reserve effects on fishes, mostly on coral reefs and in seagrass meadows (Fig. 2). The main metrics measured were spatial contrasts (i.e. inside versus outside reserves) in the abundance of species that have commercial value, or are of conservation significance (14 studies). Eight studies compared differences in the diversity and species composition of fish assemblages between reserves and fished locations.

Work on the ecological roles and functions of fishes inside and outside of reserves is centred on herbivory, either on coral reefs (six studies) (25), or in seagrass meadows (one study) (26). Enhanced grazing of algae by fishes within reserves has significant effects on benthic habitat structure of corals and the physiology of seagrass species (26, 27). The fish species most frequently identified as a functionally important herbivore is the dusky rabbitfish Siganus fuscescens. This is a harvested species (34), which varies in abundance seasonally (24), and is strongly affected by the proximity of coral reefs and seagrass meadows to other ecosystems, especially mangroves (e.g. seascape connectivity effects) (27, 35). The effects of reserves on other ecological functions (e.g. scavenging, carbon processing, predation) have not been sufficiently studied, but there is an indication that estuarine reserves might promote scavenging and carbon processing (15), whereas soft-sediment reserves do not appear to modify predation pressure (36).
Changes to ecological functions performed by fishes that are associated with reserves, are also predicted to propagate to effects on the condition of benthic ecosystems. For example, reserves that conserve both coral reefs and nearby mangroves contain more roving herbivores (22), which consume more algae (i.e. higher herbivory) and promote coral recruitment on reefs (27). Macroalgal blooms on reefs in response to high nutrient loads can smother corals in Moreton Bay (11). However, abundant herbivorous fishes rapidly consume algae in some marine reserves; a function that enhanced the resilience of protected reefs to the impacts of flood disturbance in 2011 (28). Herbivorous rabbitfish are also important herbivores in seagrass meadows (24, 37), and are more abundant in marine reserves than in similar fished locations (35), but it is not clear whether these reserve effects also modify herbivory in seagrass meadows (26). No studies in Moreton Bay have addressed questions about reserve-associated changes to ecological functions, or the condition of benthic communities for ocean beaches, mangroves, and soft-sediment ecosystems within Moreton Bay (Fig. 2).
Ecological features that shape conservation performance
A recurrent question in conservation planning is ‘What factors determine the effectiveness of marine reserves?’ (Fig. 3). In Moreton Bay, several studies have sought to identify the ecological factors that contribute most to making reserves effective. The effects of seascape context (i.e. the spatial attributes of ecosystems within heterogeneous seascapes, including their position, isolation and size)(38) for reserve performance feature prominently in the literature from Moreton Bay. Effects of seascape context on reserve performance have been examined for coral reefs (22), seagrass meadows (35), estuaries (39) and surf zones (40). Across each of these ecosystems, marine reserves that conserved prominent features and seascape connections were more effective than those that did not. For example, reserves that conserved both coral reefs and mangroves in close proximity were more effective for fishes than those that did not (Fig. 3a, b) (22). Similarly, reserves that conserve seagrass meadows, which are close to the open ocean, perform better for fishes than reserves that are farther from the sea (35). By contrast, reserves in estuaries perform poorly for fishes (i.e. abundance does not differ between reserves and fished waters) because they conserve locations with limited habitat values (i.e. narrow estuaries with weak connections to mangrove habitats and the open ocean) (39). Small reserves that protect the surf zones of ocean beaches in Moreton Bay were not found to be effective for fishes in their current positions (40). We know, however, from other regions along the Australian east coast that surf-zone reserves perform better for fishes when they conserve large areas that include both ocean beaches and rocky headlands in close proximity (40).

Moreton Bay has a distinct gradient in water quality, characterised by turbid, nutrient-rich waters towards the western and southern margins where estuaries discharge, and cleaner, oceanic waters in the east and north (11, 41). These water-quality effects influence the composition of some fish communities (23, 35). They do not, however, modify the strong effects of seascape context on fish assemblages on coral reefs, and in seagrass meadows (23, 35). By contrast, variation in water quality is particularly important in shaping the composition of benthic communities on coral reefs, and in seagrass meadows, which are frequently dominated by macroalgae in areas that receive higher nutrient concentrations (11, 41) (Fig. 3c, d). Coral reefs that are within reserves, however, recover from eutrophication more quickly than reefs that are open to fishing, because they support more herbivorous fishes (28).
The effects of fishing outside reserves, and movement of species beyond reserve boundaries, is also a crucial consideration for conservation in Moreton Bay given the spatial scale of the current reserves in the Bay (23, 42). Many fish species might migrate across the boundaries of marine reserves during tidal, diel or seasonal movements, thereby increasing their risk of being caught by fishers (43, 44), however further research into these effects needs to be conducted specifically for reserves in Moreton Bay (Fig. 3e, f). We suggest that large, highly mobile species (e.g. elasmobranchs) are particularly susceptible to the effects of fishing outside of reserves, but this prediction has not been tested in Moreton Bay. Empirical data is therefore needed to determine whether, and how, the distribution and intensity of fishing effort in Moreton Bay influences reserve performance.
Synthesis and future directions
This review demonstrates that reserves can be effective for restoring fish assemblages, key ecological functions and some key habitats in Moreton Bay. This is, however contingent upon the reserves being appropriately large for the species being protected, on the reserve being connected with nearby alternative habitats (especially if these functional linkages are also preserved), and the reserve being placed strategically along a gradient (such as riverine run-off). In unison, these findings, whilst important for Moreton Bay specifically, reflect the findings of several broader reserve effectiveness studies globally (2, 45, 46). Consequently, the effects of no-take reserves in Moreton Bay are highly variable. They can have positive effects on numerous ecosystem components in some locations, but might have little, or no, detectable effect in other settings. We found that reserves in Moreton Bay were effective for fish, ecological functions and benthic communities in some ecosystems (i.e. coral reefs, seagrass meadows), and less effective for others (i.e. surf zones, rocky reefs). Whilst the performance of marine reserves broadly across Moreton Bay is principally shaped by: (i) the level of connectivity between habitat patches, and (ii) the position of reserves along the ambient water-quality gradient, the intensity of these effects varies among ecosystems. The effects of reserves on benthic assemblages, ecological functions and resilience have only been comprehensively examined for coral reefs, and more research is needed to determine whether these reserve effects occur in other ecosystems. These types of reserve effects are complex and can be difficult to measure, partly because disturbances that occur outside reserve boundaries (e.g. eutrophication, sedimentation, pollution, fishing) also influence the composition, and trophic ecology, of fish assemblages within reserves. The magnitude of reserve effects can, therefore, vary within and among ecosystems depending on the level of external disturbance (e.g. the gradient of water-quality degradation from west-to-east across Moreton Bay), and the spatial properties of local seascapes (e.g. ecosystem context, connectivity, size).
These findings are analogous to results from other parts of the world, where the spatial connections between functionally linked habitats have been found to be principal determinants of marine reserve success in many coastal seascapes (21, 47), and where exogenic unmanaged pressures are considered as one of the most important threats to effective management of marine ecosystems worldwide (48). Because data on reserve effectiveness are lacking for some habitats, we cannot determine whether reserve effectiveness differs among ecosystems, but comparison is probably far less important than having a network of reserves that is representative, sufficiently enforced and monitored, across all ecosystem types.
Because a number of external factors (e.g. connectivity and water quality) govern the effectiveness of no-take reserves, it is important to diversify management and conservation interventions in Moreton Bay, beyond simply creating additional reserves or expanding existing ones (49). It has been demonstrated in other systems that reserve implementation is not always sufficient for conservation, and that a broader approach that tackles catchment-borne sedimentation, water quality and habitat loss is necessary to manage the full suite of impacts affecting coastal ecosystems (49, 50). For example, the success of reserves in Moreton Bay could be significantly modified by the outcomes of catchment management activities (49, 51). Consequently, one strategy could be to purposefully place some reserves in parts of the Bay where flood impacts are lower (28, 41). Numerous authors have stressed the importance of accounting for reserve size and the movement of species when designingg new reserves (52). Given our current understanding of fish movement in Moreton Bay (23, 24, 42), some existing reserves may not be large enough to encompass the daily home ranges of species that move among mangroves, seagrasses and coral reefs (22). Any new reserves should, therefore, be designed to conserve focal habitats for the species or assemblages of interest, and include all connections with adjacent habitats that these taxa use regularly for feeding, sheltering, or breeding purposes (22).
There are several sizable gaps in our understanding of reserve performance in Moreton Bay. Research effort has primarily focused on three ecosystems in the central and southern portions of Moreton Bay (i.e. coral reefs, mangroves, seagrasses), whereas the performance of reserves in other ecosystems has not been sufficiently studied. For example, subtidal soft-sediments, sandbanks and mud flats are the most widespread, and common, ecosystems in central Moreton Bay. Trawling closures can modify the epi-benthic communities of subtidal soft-sediments in the central bay (53), but it is not clear whether these effects also propagate to fish assemblages and ecological functions. Similarly, coastal reserves modify the fauna of ocean beaches, and their surf zones, in Queensland; sometimes for the better in terms of increasing targeted fish species, and other times negatively in terms of increasing invasive species along sandy beaches (40, 54, 55), but these effects have not been explicitly tested in Moreton Bay.
The effects of reserves on ecological functions and benthic communities have been reasonably well-studied on coral reefs, and in seagrass meadows, but we do not know whether these functional effects of reserves are common, or widespread, in other ecosystems. Within Moreton Bay, there has been significant focus on how reserves influence herbivory and some work to measure reserve effects on predation and scavenging, but other critical processes remain poorly studied. For example, it is not clear whether reserves in Moreton Bay also influence larval recruitment and settlement, nutrient budgets, or productivity (either primary or secondary). Because the performance of reserves usually improves over time, continued monitoring will be critical to determine whether trajectories differ among ecosystems. Finally, to determine the potential effects of fishing on reserve performance, we suggest that data are needed test how variation in fishing effort outside marine reserves correlates with fish movement across reserve boundaries. This might be particularly important for large, highly mobile species with home ranges that are larger than reserves, such as elasmobranchs (42).
Conclusions
Reserves in Moreton Bay are effective for enhancing the abundance of harvested fish species, and improving ecological functions in some ecosystems (i.e. coral reefs, seagrass meadows), but might not be particularly effective in others (i.e. estuaries, surf zones). The performance of reserves has not been studied sufficiently in many ecosystems (subtidal soft-sediments, ocean beaches, offshore reefs). The most important considerations given research to date in terms of maximising the effectiveness of reserves in Moreton Bay are how well ecosystems are spatially connected within reserve seascapes, and their resilience to variable water quality from catchment and estuarine runoff. Significant progress has been made to understand the effects of no-take reserves over the last two decades, but there are numerous gaps in our ecological knowledge of how reserves function. To better understand conservation performance in Moreton Bay, and help optimise future management decisions, studies of reserve effectiveness must be broadened to investigate how reserves shape fish assemblages and ecological functions across all ecosystems in Moreton Bay. Finally, the success of reserves in Moreton Bay could be significantly modified by the outcomes of catchment management activities that govern the water quality gradient across the Bay.
References
- Douvere F. 2008. The importance of marine spatial planning in advancing ecosystem-based sea use management. Marine Policy. 32(5):762-771. 10.1016/j.marpol.2008.03.021
- Edgar GJ, Stuart-Smith RD, Willis TJ, Kininmonth S, Baker SC, Banks S, Barrett NS, Becerro MA, Bernard AT, Berkhout J, Buxton CD, Campbell SJ, Cooper AT, Davey M, Edgar SC, Forsterra G, Galvan DE, Irigoyen AJ, Kushner DJ, Moura R, Parnell PE, Shears NT, Soler G, Strain EM, Thomson RJ. 2014. Global conservation outcomes depend on marine protected areas with five key features. Nature. 506:216-220. 10.1038/nature13022
- Gilby BL, Stevens T. 2014. Meta-analysis indicates habitat-specific alterations to primary producer and herbivore communities in marine protected areas. Global Ecology and Conservation. 2:289-299. 10.1016/j.gecco.2014.10.005
- Mumby PJ, Dahlgren CP, Harborne AR, Kappel CV, Micheli F, Brumbaugh DR, Holmes KE, Mendes JM, Broad K, Sanchirico JN, Buch K, Box S, Stoffle RW, Gill AB. 2006. Fishing, trophic cascades, and the process of grazing on coral reefs. Science. 311(5757):98-101. 10.1126/science.1121129
- Shears NT, Babcock RC, Salomon AK. 2008. Context-dependent effects of fishing: Variation in trophic cascades across environmental gradients. Ecological Applications. 18(8):1860-1873. 10.1890/07-1776.1
- Shears N, Babcock R. 2002. Marine reserves demonstrate top-down control of community structure on temperate reefs. Oecologia. 132(1):131-142. 10.1007/s00442-002-0920-x
- Edgar GJ, Barrett NS. 1997. Short term monitoring of biotic change in Tasmanian marine reserves. Journal of Experimental Marine Biology and Ecology. 213(2):261-279
- Millar RB, Willis TJ. 1999. Estimating the relative density of snapper in and around a marine reserve using a log-linear mixed-effects model. Australian & New Zealand Journal of Statistics. 41(4):383-394
- Di Lorenzo M, Claudet J, Guidetti P. 2016. Spillover from marine protected areas to adjacent fisheries has an ecological and a fishery component. Journal for Nature Conservation. 32:62-66. 10.1016/j.jnc.2016.04.004
- Russ GR, Alcala AC, Maypa AP, Calumpong HP, White AT. 2004. Marine reserve benefits local fisheries. Ecological Applications. 14(2):597-606
- Gilby BL, Maxwell PS, Tibbetts IR, Stevens T. 2015. Bottom-up factors for algal productivity outweigh no-fishing marine protected area effects in a marginal coral reef system. Ecosystems. 18(6):1056-1069
- Mumby PJ, Harborne AR, Williams J, Kappel CV, Brumbaugh DR, Micheli F, Holmes KE, Dahlgren CP, Paris CB, Blackwell PG. 2007. Trophic cascade facilitates coral recruitment in a marine reserve. Proceedings of the National Academy of Sciences of the United States of America. 104(20):8362-8367. 10.1073/pnas.0702602104
- Mumby PJ, Harborne AR. 2010. Marine reserves enhance the recovery of corals on Caribbean reefs. PLoS ONE. 5:e8657. 10.1371/journal.pone.0008657
- Clemente S, Hernández JC, Brito A. 2011. Context-dependent effects of marine protected areas on predatory interactions. Marine Ecology-Progress Series. 437:119-133
- Webley JAC. 2008. The ecology of the mud crab (Scylla serrata): Their colonisation of estuaries and role as scavengers in ecosystem processes. Griffith University. Gold Coast, Australia
- McClanahan TR. 2000. Recovery of a coral reef keystone predator, Balistapus undulatus, in East African marine parks. Biological Conservation. 94(2):191-198. 10.1016/s0006-3207(99)00176-7
- McClanahan TR, Graham NAJ, Calnan JM, MacNeil MA. 2007. Toward pristine biomass: Reef fish recovery in coral reef marine protected areas in Kenya. Ecological Applications. 17(4):1055-1067. 10.1890/06-1450
- Possingham HP, Franklin J, Wilson K, Regan TJ. 2005. The roles of spatial heterogeneity and ecological processes in conservation planning. In: Lovett GM, Jones CG, Turner MG, Weathers KC. (Eds.) Ecosystem function in heterogeneous landscapes. Springer, New York. p. 389-406. 10.1007/0-387-24091-8_19. <Go to ISI>://WOS:000235132700019
- Beger M, Grantham HS, Pressey RL, Wilson KA, Peterson EL, Dorfman D, Mumby PJ, Lourival R, Brumbaugh DR, Possingham HP. 2010. Conservation planning for connectivity across marine, freshwater, and terrestrial realms. Biological Conservation. 143(3):565-575. 10.1016/j.biocon.2009.11.006
- Huijbers CM, Schlacher TA, Schoeman DS, Olds AD, Weston MA, Connolly RM. 2015. Limited functional redundancy in vertebrate scavenger guilds fails to compensate for the loss of raptors from urbanized sandy beaches. Diversity and Distributions. 21(1):55-63. 10.1111/ddi.12282
- Olds AD, Connolly RM, Pitt KA, Pittman SJ, Maxwell PS, Huijbers CM, Moore BR, Albert S, Rissik D, Babcock RC, Schlacher TA. 2016. Quantifying the conservation value of seascape connectivity: A global synthesis. Global Ecology and Biogeography. 25:3-15. 10.1111/geb.12388
- Olds AD, Connolly RM, Pitt KA, Maxwell PS. 2012. Habitat connectivity improves reserve performance. Conservation Letters. 5(1):56-63. 10.1111/j.1755-263X.2011.00204.x
- Gilby BL, Tibbetts IR, Olds AD, Maxwell PS, Stevens T. 2016. Seascape context and predators override water quality effects on inshore coral reef fish communities. Coral Reefs. 35(3):979-990
- Gilby BL, Tibbetts IR, Stevens T. 2016. Low functional redundancy and high variability in Sargassum browsing fish populations in a subtropical reef system. Marine and Freshwater Research. 68:331-341
- Yabsley NA, Olds AD, Connolly RM, Martin TSH, Gilby BL, Maxwell PS, Huijbers CM, Schoeman DS, Schlacher TA. 2015. Resource type modifies the effects of reserves and connectivity on ecological functions. Journal of Animal Ecology. 82(2):437-444
- Henderson CJ, Stevens T, Olds AD, Gilby BL, Warnken J, Lee SY. 2017. Optimising seagrass conservation for ecological functions. Ecosystems.In press
- Olds AD, Pitt KA, Maxwell PS, Connolly RM. 2012. Synergistic effects of reserves and connectivity on ecological resilience. Journal of Applied Ecology. 49(6):1195-1203
- Olds AD, Pitt KA, Maxwell PS, Babcock RC, Rissik D, Connolly RM. 2014. Marine reserves help coastal systems cope with extreme weather. Global Change Biology. 20:3050-3058
- Humphries AT, McQuaid CD, McClanahan TR. 2015. Context-dependent diversity-effects of seaweed consumption on coral reefs in Kenya. PLoS ONE. 10(12):e0144204. 10.1371/journal.pone.0144204
- Edgar GJ, Barrett NS, Stuart-Smith RD. 2009. Exploited reefs protected from fishing transform over decades into conservation features otherwise absent from seascapes. Ecological Applications. 19(8):1967-1974
- Helen Ross, Rissik D, Jones N, Witt K, Pinner B, Shaw S. 2019. Managing for the multiple uses and values of Moreton Bay and its catchments. In: Tibbetts IR, Rothlisberg PC, Neil DT, Homburg TA, Brewer DT, Arthington AH. (Eds.) Moreton Bay Quandamooka & Catchment: Past, present and future. The Moreton Bay Foundation, Brisbane, Australia. Available from: https://moretonbayfoundation.org/
- Pillans S, Pillans RD, Johnstone RW, Kraft PG, Haywood MDE, Possingham HP. 2005. Effects of marine reserve protection on the mud crab Scylla serrata in a sex-biased fishery in subtropical Australia. Marine Ecology-Progress Series. 295:201-213
- Pillans S, Ortiz J-C, Pillans RD, Possingham HP. 2007. The impact of marine reserves on nekton diversity and community composition in subtropical eastern Australia. Biological Conservation. 136(3):455-469. 10.1016/j.biocon.2006.12.018
- Terres MA, Lawrence E, Hosack GR, Haywood MD, Babcock RC. 2015. Assessing habitat use by snapper (Chrysophrys auratus) from baited underwater video data in a coastal marine park. PLoS ONE. 10(8):e0136799. 10.1371/journal.pone.0136799
- Tibbetts IR, Townsend KA. 2010. The abundance, biomass and size of macrograzers on reefs in Moreton Bay, Queensland. Memoirs of the Queensland Museum. 54(3):373-384
- Henderson CJ, Olds AD, Lee SY, Gilby BL, Maxwell PS, Connolly RM, Stevens T. 2017. Marine reserves and seascape context shape fish assemblages in seagrass ecosystems. Marine Ecology Progress Series. 566:135-144
- Connolly RM, Pitt KA, Rissik D, Babcock RC, Olds AD, Maxwell PS, Burfeind DD, Beattie CL. 2012. Influence of marine protected areas on ecosystem resilience and ecological processes. Final ARC linkage report to Queensland Department of Science, Information Technology, Innovation and the Arts.
- Ebrahim A, Olds AD, Maxwell PS, Pitt KA, Burfeind DD, Connolly RM. 2014. Herbivory in a subtropical seagrass ecosystem: Separating the functional role of different grazers. Marine Ecology Progress Series. 511:83-91. 10.3354/meps10901
- Pittman SJ, Kneib RT, Simenstad CA. 2011. Practicing coastal seascape ecology. Marine Ecology Progress Series. 427:187-190. 10.3354/meps09139
- Gilby BL, Olds AD, Yabsley NA, Maxwell PS, Connolly RM, Schlacher TA. 2017. Enhancing the performance of marine reserves in estuaries: Just add water. Biological Conservation. 210:1-7
- Ortodossi N, Olds AD, Gilby BL, Connolly RM, Schlacher TA. 2019. Effects of seascape connectivity on reserve performance along exposed coastlines. Conservation Biology. 33:580-589
- Maxwell PS, Pitt KA, Burfeind DD, Olds AD, Babcock RC, Connolly RM. 2014. Phenotypic plasticity promotes persistence following severe events: Physiological and morphological responses of seagrass to flooding. Journal of Ecology. 102(1):54-64. 10.1111/1365-2745.12167
- Henderson CJ, Stevens T, Gilby BL, Lee SY. 2017. Spatial conservation of large mobile elasmobranchs requires an understanding of spatio-temporal seascape utilisation. ICES Journal of Marine Science. 72(2):553-561
- Kramer DL, Chapman MR. 1999. Implications of fish home range size and relocation for marine reserve function. Environmental Biology of Fishes. 55:65-79
- Weeks R, Green AL, Joseph E, Peterson N, Terk E, Bode M. 2017. Using reef fish movement to inform marine reserve design. Journal of Applied Ecology. 54(1):145-152. 10.1111/1365-2664.12736
- Halpern BS. 2003. The impact of marine reserves: Do reserves work and does reserve size matter? Ecological Applications. 13(1):S117-S137
- Huijbers CM, Connolly RM, Pitt KA, Schoeman DS, Schlacher TA, Burfeind DD, Steele C, Olds AD, Maxwell PS, Babcock RC, Rissik D. 2015. Conservation benefits of marine reserves are undiminished near coastal rivers and cities. Conservation Letters. 8(5):312-319. 10.1111/conl.12128
- Olds AD, Albert S, Maxwell PS, Pitt KA, Connolly RM. 2013. Mangrove-reef connectivity promotes the effectiveness of marine reserves across the western Pacific. Global Ecology and Biogeography. 22(9):1040-1049. 10.1111/geb.12072
- Elliott M. 2011. Marine science and management means tackling exogenic unmanaged pressures and endogenic managed pressures – a numbered guide. Marine Pollution Bulletin. 62:651-655
- Gilby BL, Olds AD, Connolly RM, Stevens T, Henderson CJ, Maxwell PS, Tibbetts IR, Schoeman DS, Rissik D, Schlacher TA. 2016. Optimising land-sea management for inshore coral reefs. PLoS ONE. 11(10):e0164934. 10.1371/journal.pone.0164934
- Margules CR, Pressey RL. 2000. Systematic conservation planning. Nature. 405(6783):243-253. 10.1038/35012251
- Saxton NE, Olley JM, Smith S, Ward DP, Rose CW. 2012. Gully erosion in sub-tropical south-east Queensland, Australia. Geomorphology. 173:80-87. 10.1016/j.geomorph.2012.05.030
- Vandeperre F, Higgins RM, Sanchez-Meca J, Maynou F, Goni R, Martin-Sosa P, Perez-Ruzafa A, Afonso P, Bertocci I, Crec’hriou R, D’Anna G, Dimech M, Dorta C, Esparza O, Falcon JM, Forcada A, Guala I, Le Direach L, Marcos C, Ojeda-Martinez C, Pipitone C, Schembri PJ, Stelzenmuller V, Stobart B, Santos RS. 2011. Effects of no-take area size and age of marine protected areas on fisheries yields: A meta-analytical approach. Fish and Fisheries. 12(4):412-426. 10.1111/j.1467-2979.2010.00401.x
- Stevens T, Richmond SJ, Wiliams E, Rissik D, Suddrey C. 2014. Effects of cessation of trawling activities within Moreton Bay Marine Park on benthic assemblages. Report to the Queensland Department of Science, Information Technology, Innovation and the Arts. Griffith University. Brisbane, Australia
- Schlacher TA, Weston MA, Lynn D, Schoeman DS, Huijbers CM, Olds AD, Masters S, Connolly RM. 2015. Conservation gone to the dogs: When canids rule the beach in small coastal reserves. Biodiversity and Conservation. 24:493-509
- Sheppard N, Pitt KA, Schlacher TA. 2009. Sub-lethal effects of off-road vehicles (ORVS) on surf clams on sandy beaches. Journal of Experimental Marine Biology and Ecology. 380(1-2):113-118. 10.1016/j.jembe.2009.09.009
- Olds AD, Frohloff BA, Gilby BL, Connolly RM, Yabsley NA, Maxwell PS, Schlacher TA. 2018. Urbanisation supplements ecosystem functioning in disturbed estuaries. Ecography.41(12):2104-13
